

Engineering
Precision Medicines
Clinical Stage.
Platform Driven Products.
Nanomerics is an award-winning clinical-stage biopharmaceutical company developing precision medicines for ophthalmology and CNS conditions. Founded in 2010 by world-leading scientists at UCL, our proprietary Molecular Envelope Technology (MET) platform enhances and repurposes known drugs — getting more drug to the site of action, with improved activity and reduced side effects.
Spun out from UCL, Strathclyde and Glasgow with deep roots in academic science and a track record in clinical translation. The platform has been validated across multiple drug classes, earning the King's Award for Enterprise and the RSC Emerging Technologies Prize.
Learn more about partnering →Spun out of UCL, University of Glasgow and Strathclyde by Prof Ijeoma Uchegbu and Prof Andreas Schätzlein to commercialise proprietary molecular engineering know-how
Molecular Envelope Technology validated across multiple drug classes — winner of the King's Award for Enterprise: Innovation 2024 and RSC Emerging Technologies Prize
Two CNS-targeted products out-licensed — Envelta™ (NB127), a non-addictive analgesic for nose-to-brain delivery, partnered with Virpax Pharmaceuticals. NIH NCATS collaboration initiated to advance MET platform for CNS indications.
Non-viral gene delivery platform achieves 50–55% gene knockdown in the cerebral cortex via intranasal administration with 1,830× lower cytotoxicity than the gold standard
OC134 Phase I clinical trial met all primary endpoints. Both OC134 and the MET platform were well tolerated in healthy volunteers — no adverse events of moderate or severe severity (March 2025)
Leadership & Advisory Board

Andreas G Schätzlein
Dr med vetStart-up team IDEA Ag. Thirty years of experience in medicines development and commercialisation with multiple deals negotiated, moving repeatedly between academia and industry. Professor of Translational Therapeutics, University College London. h-index 43.

Dame Ijeoma Uchegbu
PhD, FMedSci, HonFRSCMulti-award-winning nanotechnology scientist and lead inventor of the MET platform. Professor of Pharmaceutical Nanoscience, University College London. Board member, Wellcome Trust. President, Wolfson College Cambridge. h-index 56.

Andrea Mica
MScOver 30 years of experience in technology commercialisation and investment. Director at Oxford Technology SEIS/EIS Investment.

Alan Bye
PhDOver 40 years in senior pharmaceutical industry roles (Wellcome, Upjohn, Glaxo). Former VP Discovery Medicine for the Psychiatry Centre of Excellence at GSK. Led more than 300 first-in-human studies and the successful launch of over 40 commercial products.
Scientific Advisory Board
Prof. Sajjad Ahmad
MB BS PhD FRCOphthConsultant Ophthalmic Surgeon, Moorfields Eye Hospital; Professor, UCL Institute of Ophthalmology
Prof. Jon K Dart
BA BM BCh DM FRCS FRCOphthConsultant Ophthalmic Surgeon, Moorfields Eye Hospital; Professor, UCL Institute of Ophthalmology
Dr Hari Jayaram
MA MSc PhD FRCSEd FRCOphthConsultant Ophthalmic Surgeon & Director of Glaucoma Service, Moorfields Eye Hospital; Associate Professor, UCL Institute of Ophthalmology
Prof. Shalesh Kaushal
BS MD PhDEmeritus Chair Professor, University of Massachusetts Ophthalmic Centre; Principal, Comprehensive Retina Consultants, Florida, USA
Core Team

Pedro Margarido
MBAHead of Operations

Ilona Kubajewska
PhDDirector of Preclinical R&D

Asya Petkova
PhDDirector Clinical Trial Projects

Rui Manuel Jesus Lopes
PhDSenior Director and Head of Laboratories
Molecular Engineered
Materials
Nanomerics is a world leader in Molecular Engineered Materials — biocompatible polymer nanotechnologies that overcome the delivery barriers limiting conventional medicines across ophthalmology, CNS, and gene therapy.
Molecular Envelope Technology
Biocompatible polymer nanoparticles that package hydrophobic drugs and deliver them across epithelial barriers — to the front and back of the eye, and to the CNS via the oral or nasal route. Non-irritant, aqueous, and engineered for patient compliance.
- 5–10× enhanced ocular penetration
- Phase I complete — primary endpoints met
- Compatible with small molecules & peptides
METfect
Non-viral, intranasal siRNA and pDNA delivery to the brain. METfect nanoparticles are deposited in the olfactory region and transported directly to multiple brain regions via the olfactory and perivascular pathways — bypassing the blood-brain barrier without injection.
- 50–55% CNS gene knockdown in vivo
- Self-administered nasal powder · re-dosable
- siRNA, pDNA, and gene powder compatible
Molecular Envelope
Technology
An award-winning Active Excipient platform that engineers biocompatible polymer nanoparticles to dramatically increase the amount of drug available at the disease site.
Not Your Usual Nanoparticle
A polymer matrix — not a micelle with a hydrophobic core
MET is a synthetically controlled polymer that self-assembles into sub‑100 nm nanoparticles. Unlike conventional micelles — which form a core‑shell structure with a hydrophobic interior — MET creates a matrix architecture through non‑covalent cross‑linking of multiple amphiphilic groups on a single polymer chain.
This matters because in a micelle the hydrophobic core is an ideal environment for the drug — there is no thermodynamic incentive for the API to leave. The result: poor release and limited tissue penetration. In the MET matrix, drugs sit in dynamic binding pockets — a ‘Goldilocks’ environment that accommodates hydrophobic compounds readily, but only while the pockets exist.
When the positively charged particle binds to a negatively charged cell surface, electrostatic forces exceed the forces maintaining the binding pockets. The pockets collapse, and the drug is released at high local concentration directly at the cell membrane — driving tissue penetration rather than trapping the API indefinitely.
Dynamic binding pockets. Drug released on cell contact.
Hydrophobic core sequesters drug. No release trigger.
Stability Means Safety
Multiple amphiphilic groups in one molecule create extremely stable self‑assemblies with ultra‑low CMC — orders of magnitude more stable and safer than other self‑assembling systems.
10 GCPQ chains forming an intertwined matrix · 200 ns MD
Why ‘Molecular Envelope’
Polymer chains change conformation to maximise drug binding — enveloping individual API molecules throughout the matrix
Unlike surface adsorption or passive entrapment, MET polymers actively adapt their conformation around each drug molecule. Intra‑ and inter‑polymer binding pockets form dynamically, accommodating hydrophobic compounds through induced‑fit interactions — enabling up to 50% drug loading compared to 1–10% with conventional technologies.
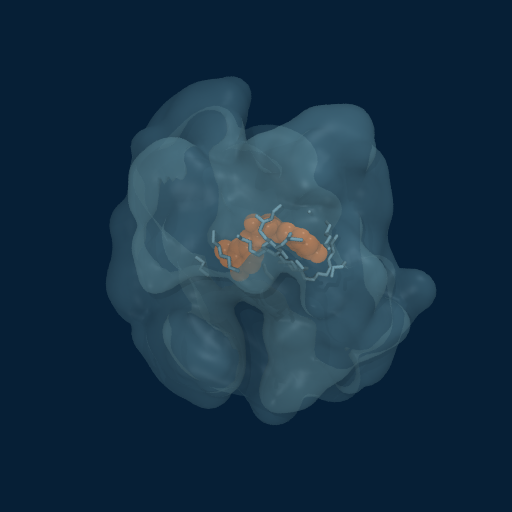
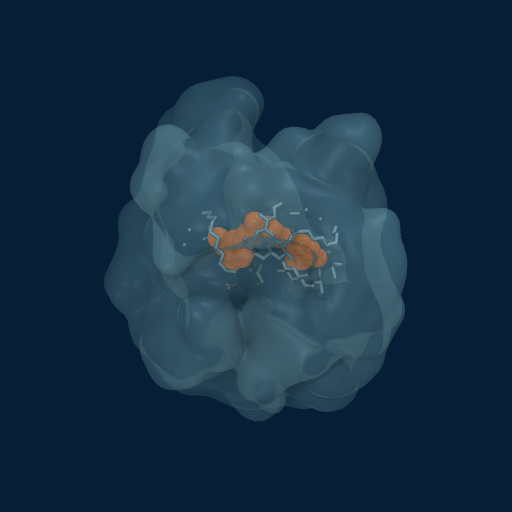
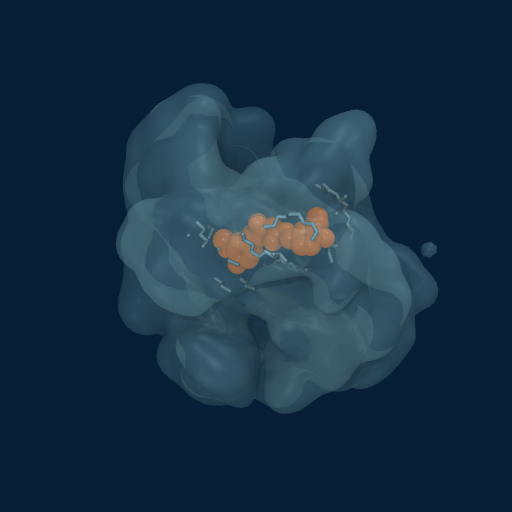
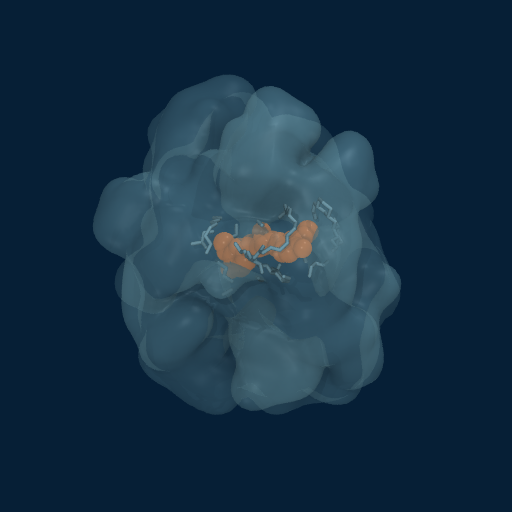
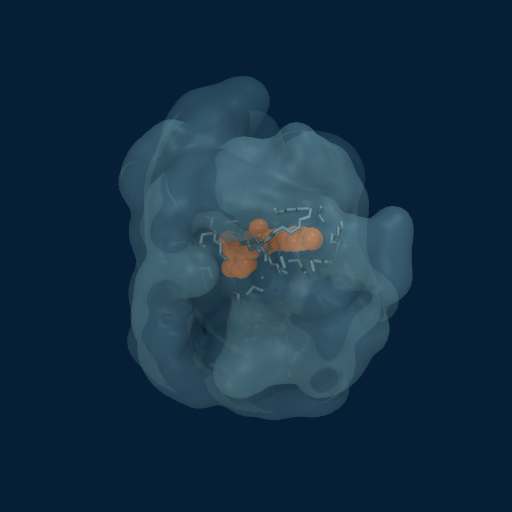
Drug Enveloped by Polymer Matrix
Curcumin (orange) progressively enveloped by GCPQ polymer chains (translucent surface). Multiple chains contribute to the dynamic binding pocket.
Mechanism of Action
From self‑assembly to drug release — a charge‑triggered delivery mechanism
Mechanism of Action
MET nanoparticles envelope drug molecules and release them at the cell surface via charge-triggered conformational change
Self-Assembly
Individual MET polymer chains spontaneously self-assemble into stable matrix nanoparticles at ultra-low critical micellar concentration (CMC ~20 μM) — orders of magnitude more stable than conventional self-assembling systems.
Drug Loading
Polymer chains change conformation to form dynamic binding pockets that envelope individual drug molecules throughout the matrix — enabling up to 50% drug loading vs 1–10% with conventional technologies.
Aqueous Transport
Cationic nanoparticles carry a high drug payload in a water-like aqueous formulation. Low dynamic viscosity (<5 mPa·s) preserves visual acuity with no burning or stinging.
Cell Binding & Conformational Change
Electrostatic forces at the cell surface exceed the dynamic pocket-forming forces. Polymer chains flatten against the membrane, losing the ability to envelope drug molecules.
Drug Release & Tissue Penetration
Released drug creates a high local concentration at the cell membrane, facilitating partitioning into cells and sustained tissue retention without systemic exposure.
Comparative Efficiency
Head‑to‑head comparison of ocular delivery technologies using published CsA data
The rabbit eye is the recognised preclinical model for human ocular pharmacokinetics — drug concentrations in the rabbit are predictive of human eye concentrations, allowing direct comparison of eye drop technologies.
Using cyclosporine A (CsA) — the drug for which comparable published data exist across all major platforms — MET penetration enhancement dramatically outperforms competing technologies.
2–3× more drug to cornea than Santen’s cationic emulsion
4–10× more drug to cornea than Restasis (Allergan)
7–11× more drug to conjunctiva than NOVA22007 or Restasis
Cmax (ng/g per μg dosed) · Rabbit model · Published data
Engineered Supramolecular Self-assembly
Biocompatible polymers are tailored to form nano-scaled containers that package hydrophobic drugs in aqueous media. The highly dynamic encapsulation allows efficient drug loading and rapid release at the target barrier.
Ocular & CNS Delivery
MET delivers drugs to the front and back of the eye via non-irritant aqueous eye drops, and to the brain via the nose-to-brain route — bypassing barriers that defeat conventional formulations.
Broad Platform Applicability
Compatible with low molecular weight hydrophobic drugs and peptides across ophthalmic, nose-to-brain CNS, oral, and subcutaneous routes.
METfect
Nose-to-Brain
A non-viral, non-invasive gene delivery platform that encapsulates siRNA and pDNA in biodegradable polymeric nanoparticles for direct CNS access via the olfactory pathway.
Nose-to-Brain Delivery
Intranasal administration deposits METfect nanoparticles in the olfactory region, bypassing the blood-brain barrier via olfactory and perivascular pathways to achieve broad CNS distribution.
Payload Flexibility
A single platform compatible with siRNA, pDNA, and gene powder formats. Spray-dried nano-in-micro particles provide room-temperature stability for 3+ months, enabling scalable manufacturing.
Re-dosable Gene Medicine
Unlike viral vectors limited by immunogenicity and a single-administration paradigm, METfect nanoparticles are re-dosable — enabling chronic disease management with a patient-friendly nasal spray.
Gene Powder — First Published Polymeric Gene Dry Powder
Spray-dried METfect nano-in-micro gene powder provides room-temperature stability for 3+ months — enabling scalable manufacturing and cold-chain-free distribution for global access.
Our Pipeline
Four platform-enabled portfolios spanning ophthalmology, CNS, oral delivery, and gene delivery.
Press & Announcements
 Milestone
Milestone Nanomerics Secures US Patent Extending MET Platform Protection to the 2040s
Read more → Media
Media Chain Reaction: Dame Ijeoma Uchegbu Announces Debut Popular Science Book
Read more → Media
Media BBC World Service Outlook: 'One Suitcase, Three Kids — A Broke Scientist Pursues a Dream'
Read more →In the Media
BBC Radio
Let's Build Together
We are actively seeking development and commercialisation partners for our pipeline programmes. If you share our commitment to addressing unmet medical need, we'd welcome the conversation.
bd@nanomerics.com
